CRISPR, the angel and the demon
The angel thought that
CRISPRshould become his best friend. Although he had not always been very loyal, the “others”, those obsolete
genome engineeringtechnologies, no longer seemed to interest him. CRISPR attracted his attention as a “fantastic invention” of nature serving man’s designs. Its inventors would certainly deserve a Nobel prize for this entity which seemed perfect to help them reconstruct life for the purposes of biotechnologists. Perfect, not so much, thought the demon, who was already dreaming of exploiting all the flaws that the
laws of naturecan also create, to divert and impair the greatest human technologies. Angel and demon are
two faces of any breakthrough technology. What about the different approaches currently competing for editing genomes? [bloc_image]

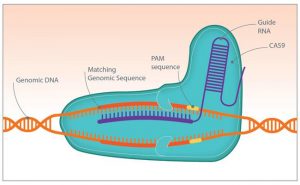
[/bloc_image]
(Credits: http://www.genengnews.com/bioperspectives/ & http://www.observatoriobioetica.org)
Targeted genome engineering is currently positioned as the cornerstone of biotechnology, whether the target is the bio-based production of defined chemical compounds, of energy or food resources, or biomedical applications. Whatever the objective, the editing of a genome, whether it is a question of introducing, modifying, or suppressing genes, involves a natural mechanism, namely
recombination, which uses the natural repair capabilities of organisms by diverting them. The problem is that these natural
mechanismsare on the one hand thoroughly
regulated, which sometimes makes them inefficient when we seek to divert them, and on the other hand
imperfect, with potential errors leading to uncontrolled genetic modifications that can have unintended or even intolerable consequences, for instance in gene therapy. The secret consisted in favouring and targeting the natural recombination mechanisms by not giving the organism the choice to do otherwise. For this, we first
breakthe genome’s
DNAusing a molecular tool whose main characteristic has to be its
specificity. This specificity must be
adjustableto target a unique sequence chosen at will. The interest of introducing a break in the genome is that a
targeted repairbecomes indispensable to the organism’s survival and that the easiest way to achieve it is to implement a homologous recombination event with a
synthetic DNAfragment, which will also enable the desired genetic modifications. This brings the problem back to having specific
molecular scissorsat one’s disposal. Many technologies (the others) have succeeded each other. Historically, natural enzymes, the
meganucleaseswhich recognize long, specific DNA sequences, were first used. However, this system did not allow one to easily choose the engineering site which was predefined by the enzyme’s natural specificity. In the following generation, biotechnologists were able to make these natural enzymes “evolve” to give them new specificities. Nevertheless, these chimeras were long and complex to design case-by-case, and often lacked efficiency although their specificity was generally good. A first breakthrough appeared with the development of modular meganucleases (
Zinc fingerand
TALENenzymes) whose specificity could be rationally designed by combining a chain of recognition modules with a catalytic cleavage domain. These enzymes, in particular the TALENs, were faster and much less costly to design, but their production however involved quite sophisticated technology, not always directly accessible to researchers who often had to have them custom-made by specialized companies. The next conceptual breakthrough led to the famous CRISPR (formally
CRISPR-CAS9) where the recognition depended not on a protein but on a guide RNA that just had to be changed to change at will the specificity. This RNA was itself expressed from DNA which is very easy and quick to chemically synthesize. CRISPR rapidly appeared as a “nobelisable” and “
almost perfect” technology for genome engineering. However, this should be
put into perspective: its specificity often does not equal that enabled by meganucleases or TALENs. Furthermore, the latter could be used by injecting within the cells to be transformed not a DNA or/and RNA but the proteins themselves, which reduced accordingly the probability
of parasite eventsresulting from the undesirable integration of foreign DNA. Moreover, CRISPR, like the other nucleases, only achieves the
first stage of the recombination, leaving open to the “natural demons” the possibility of exploiting a whole collection of other parasite events during the repair phase. So, there is a wide
scope for improvement, in particular for devices to synchronize break and repair within a same complex. However, CRISPR remains currently the best tool of
genetic technologistspending the next step. The latter could come from the use of a cleavage complex using a guide DNA and no longer an RNA and for which we are promised a higher specificity, as recently published. Nevertheless, these new tools make targeted genome engineering easily accessible to all. It will probably remain for a long time a major battlefield between our angels and our demons (and their advocates) with
inescapable ethical aspectsif we consider for example stem cell or human embryo engineering.
More information
- https://en.wikipedia.org/wiki/Genome_editing
- TALEN and CRISPR/Cas Genome Editing Systems: Tools of Discovery. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4207558/
- Meganucleases and Other Tools for Targeted Genome Engineering: Perspectives and Challenges for Gene Therapy. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3267165/
- Exploring the potential of genome editing CRISPR-Cas9 technology. Gene. 2016 Nov 8. pii: S0378-1119(16)30886-1. doi: 10.1016/j.gene.2016.11.008.


